by Erowid
Thujone is a stuctural isomer of both camphor and menthol. However, the molecular geometries of these three substances are so different that it is unlikely they operate on the same receptor sites (del Castillo et al., 1975).
| NAME : | alpha-thujone |
| CHEMICAL NAME : | 4-Methyl-1-(1-methylethyl)bicyclo[3.1.0]hexan-3-one; 3-thujanone |
| ALTERNATE CHEMICAL NAMES : | (-)-Isothujone; (1S, 4R, 5R)-(-)-3-thujanone; thujon; thujone; (-)-thujone |
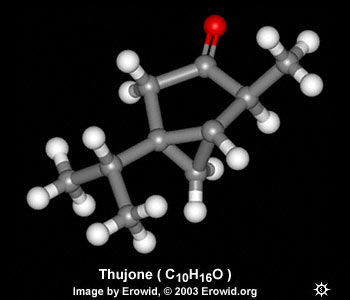
| CHEMICAL FORMULA | C10H16O |
| MOLECULAR WEIGHT | 152.24 |
| MELTING POINT | |
| BOILING POINT | 201°C |
| CAS # | 546-80-5 |
Merck Entry
9533. Thujone.
4-Methyl-1-(1-methylethyl)bicyclo[3.1.0]hexan-3-one; 3-thujanone. .C10H16O; .mol wt 152.24. .C 78.90%, H 10.59%, O 10.51%. . A constituent of many essential oils; present in thuja, etc. Equilibrium mixture contains 33% .alpha.-thujone and 67% .beta.-thujone: Eastman, Winn, J. Am. Chem. Soc. 82, 5908 (1960). .alpha.- and .beta.-Thujones differ only in the stereochemistry of the 4- methyl group. Conformation: Hach et al., Tetrahedron Letters 1970, 3175. Chemistry: J. P. Kutney et al., Bioorg. Chem. 7, 289 (1978); eidem, Can. J. Chem. 57, 3145 (1979); 58, 2641 (1980). Toxicity study: K. C. Rice, R. S. Wilson, J. Med. Chem. 19, 1054 (l976). Review: J. L. Simonsen, The Terpenes vol. II (University Press, Cambridge, 1949) pp 32-52.
Colorless or almost colorless liquid. uv max (isooctane): 300 nm (.epsilon. 23). Practically insol in water. Sol in alc and many other organic solvents. LD50 s.c. in mice: 134.2 mg/kg (Rice, Wilson).
Caution: Ingestion may cause convulsions.
l-Form, ..alpha.-thujone.. ..bp17 83.8-84.1 deg. d425 0.9109. nD15 1.4490. [.alpha.]D20 -19.2 deg. LD50 s.c. in mice: 87.5 mg/kg (Rice, Wilson).
d-Form, .d-isothujone., .beta.-thujone.. ..bp17 85.7-86.2 deg. d425 0.9135. nD25 1.4500. [.alpha.]D15 +72.5 deg. LD50 s.c. in mice: 442.2 mg/kg (Rice, Wilson).
Chemical Abstract of Thujone
by Michael Albert-Puleo
Thujone, a terpene-like ketone, is termed 3 thujamone or 3 sabinone in IUPAC nomenclature. Absinthol, tanacetone, and salvinol are terms which have been applied to the thujone extracted from Aremesia absinthium, Tanacetum vulgare, and Salvia officinalis, respectively. Thujone is a colorless oil with the following physical constants: B.p. = 199-200 C.: M.W. = 152.23: Specific Gravity = 0.915 - 0.919 at C. It is soluble in alcohol, ether, and chloroform, and insoluble in water.
The odor of thujone is said to resemble that of menthol. It is isomeric with camphor, another CNS stimulant, although its chemical structure is rather unlike camphor. Thujone can be isolated from the natural oils with bisulphate, or via fractional distillation and crystalization, and has been synthesized by man.
In nature, thujone exists as two forms, levoratory a-thujone, and dextrorotatory b-thujone, the former occuring in thuja and sage, the latter in tansy and wormwood. [Erowid note: Both alpha- and beta-thujone have been isolated from A. artemisia, though beta-thujone occurs in greater quantitiy (Sacco and Chialva, 1988)]. The work of Short and Read has shown that a-thujone and b-thunoe are in fact mixtures of the diastereoisomers l-thujone and d-isothujone in dynamic equilibrium.
- from Conrad III B. Absinthe; History in a Bottle. Chronicle Books. 1988.
OFF-SITE ABSINTHE RESOURCES
Thujone.info: Accurate, authoritative information about thujone
Chemistry & Pharmacology Info from a College Paper


