
#13 BOB
beta-METHOXY-2C-B; 4-BROMO-2,5-beta-TRIMETHOXYPHENETHYLAMINE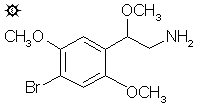
|
| [3D .mol structure] |
A solution of LAH (15 mL of 1 M solution in THF) was diluted with an equal volume of anhydrous THF, and cooled (under He) to 0 °C with an external ice bath. With good stirring there was added 0.38 mL 100% H2SO4 dropwise, to minimize charring. This was followed by the addition of 1.0 g 1-(4-bromo-2,5-dimethoxyphenyl)-1-methoxy-2-nitroethane as a solid over the course of 5 min. After an hour of stirring at 0 °C, the temperature was brought up to a gentle reflux on the steam bath for 30 min. There was no vigorous exothermic reaction seen, unlike that with the syntheses of BOD, BOH and BOM. The reaction mixture was cooled again to 0 °C, and the excess hydride was destroyed by the cautious addition of IPA. This was followed by sufficent dilute aqueous NaOH to give a white granular character to the oxides, and to assure that the reaction mixture was basic. The reaction mixture was filtered, and the filter cake washed first with THF fol-lowed by IPA. The combined filtrate and washings were stripped of solvent under vacuum and dissolved in dilute H2SO4, with the apparent generation of yellow solids. This was washed with 2x50 mL CH2Cl2, and the aqueous phase made basic with NaOH. This was extracted with 2x50 mL CH2Cl2, and the pooled extracts were stripped of solvent under vacuum. The residue was distilled at 130-150 °C at 0.2 mm/Hg to give 0.2 g of product as a clear white oil. This fraction was dissolved in 10 mL IPA, and neutralized with 4 drops concentrated HCl. The addition of 30 mL anhydrous Et2O allowed the formation of 4-bromo-2,5,beta-trimethoxyphenethylamine hydrochloride (BOB) as a fine white crystalline product. This was removed by filtration, washed with Et2O, and air dried. There was obtained 0.1 g white crystals with a mp of 187-188 °C. Anal. (C11H17BrClNO3) C,H.
DOSAGE: 10 - 20 mg.
DURATION: 10 - 20 h.
QUALITATIVE COMMENTS: (with 10 mg) I don't know if it was me this day, or if it was the chemical, but I got into a granddaddy of a paranoid, sociopathic snit, without feeling and without emotion. I was indifferent to everything. Later on, there was some improvement, with body tingling (good, I'm pretty sure) and a sense of awareness (good, I guess) but I still canceled my evening dinner company. All in all, pretty negative.
(with 10 mg) I had to get away and into myself, so I weeded in the vegetable garden for almost an hour. Then I lay down in the bedroom, and enjoyed a magnificent vegetable garden, in Southern France, in my mind's eye. An extraordinary zucchini. And the weeds had all been magically pulled. In another couple of hours a neurological over-stimulation became apparent, and I spent the rest of the day defending myself. In the evening, I took 100 milligrams phenobarbital which seemed to smooth things just enough. Too bad. Nice material, otherwise.
(with 15 mg) The erotic was lustful, but at the critical moment of orgasm, the question of neurological stability became quite apparent. Does one really let go? Everything seemed a bit irritable. The tinnitus was quite bad, but the excitement of the rich altered place I was in was certainly worth it all. Through the rest of the day, I became aware of how tired I was, and how much I wanted to sleep, and yet how scared I was to give myself over to sleep. Could I trust the body to its own devices without me as an overseeing caretaker? Let's risk it. I slept. The next day there was a memory of this turmoil. Clearly the first part of the experience might have been hard to define, but it was quite positive. But the last part makes it not really worth while.
EXTENSIONS AND COMMENTARY: This compound, BOB, is the most potent of the BOX series. And yet, as with all of the members of this family, there are overtones of physical concern, and of some worry as to the integrity of the body. There may well be a separation of activity with the two optical isomers, but there is not a tremendous push to explore this particular family much further. They can't all be winners, I guess. What would be the activities of compounds with a sulfur instead of an oxygen at the beta-oxygen position? What would be the nature of action if there were an alpha-methyl group, making all of these into amphetamine derivatives? Or what about both a sulfur and a methyl group? And what about the isomers that are intrinsic to all of this, the threo- and the erythro- and the "D's" and the "L's"? All this is terra incognita, and must someday be looked into. It is chemically simple, and pharmacologically provocative. Someone, somewhere, someday, answer these questions!
| [ |
[Main Index] | [Forward |

