
#106 MDE
MDEA; EVE; N-ETHYL-MDA; 3,4-METHYLENEDIOXY-N-ETHYLAMPHETAMINE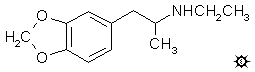
|
|
[3D .jpg image] [3D .mol structure] |
A stirred suspension of 4.8 g LAH in 400 mL anhydrous THF was brought up to a reflux, and then treated with a solution of 5.0 g of the impure N-acetyl-3,4-methylenedioxyamphetamine in 20 mL anhydrous THF. Reflux conditions were maintained for 3 days, and then after cooling in an ice bath, the excess hydride was destroyed with the careful addition of H2O. The 4.8 mL H2O (in a little THF) was followed with 4.8 mL of 15% NaOH, and finally an additional 15 mL H2O. The white, granular, basic mass of inorganic salts was removed by filtration, the filter cake washed with additional THF, and the combined filtrate and washings stripped of solvent under vacuum. The residue was dissolved in 20 mL IPA, made acidic with 40 drops of concentrated HCl, and diluted with 150 mL anhydrous Et2O. The crystalline product was removed by filtration, washed with 80% Et2O (containing IPA) followed by Et2O itself, and then air dried to provide 3.0 g of 3,4-methylenedioxy-N-ethylamphetamine hydrochloride (MDE) as fine white crystals with a mp of 198-199 °C.
(from 3,4-methylenedioxyphenylacetone with aluminum amalgam) To 40 g of thin aluminum foil cut in 1 inch squares (in a 2 L wide mouth Erlenmeyer flask) there was added 1400 mL H2O containing 1 g mercuric chloride. Amalgamation was allowed to proceed until there was the evolution of fine bubbles, the formation of a light grey precipitate, and the appearance of occasional silvery spots on the surface of the aluminum. This takes between 15 and 30 min depending on the freshness of the surfaces and the temperature of the H2O. The H2O was removed by decantation, and the aluminum was washed with 2x1400 mL of fresh H2O. The residual H2O was removed as thoroughly as possible by shaking, and there was added, in succession and with swirling, 72.5 g ethylamine hydrochloride dissolved in 60 mL warm H2O, 180 mL IPA, 145 mL 25% NaOH, 53 g 3,4-methylenedioxy-phenylacetone (see under MDMA for its preparation), and finally 350 mL IPA. The exothermic reaction was kept below 60 °C with occasional immersion into cold water and, when it was thermally stable, it was allowed to stand until it had returned to room temperature and all the insolubles settled to the bottom as a grey sludge. The clear yellow overhead was decanted and the sludge removed by filtration and washed with MeOH. The combined decantation, mother liquors, and washes, were stripped of solvent under vacuum, the residue suspended in 1500 ml of H2O, and sufficient HCl added to make the phase distinctly acidic. This was then washed with 2x100 mL CH2Cl2, made basic with 25% NaOH, and extracted with 3x100 mL of CH2Cl2. After removal of the solvent from the combined extracts, there remained 59.5 g of an amber oil which was distilled at 145-150 °C at 0.5 mm/Hg, producing 40.3 g of an off-white oil. This was dissolved in 600 mL IPA, neutralized with about 20 mL of concentrated HCl and then treated with 300 mL anhydrous Et2O. After filtering off the white crystals, washing with a IPA/Et2O (2:1) mixture, with Et2O and air drying, the final 3,4-methylenedioxy-N-ethylamphetamine hydrochloride (MDE) weighed 37.4 g.
(from 3,4-methylenedioxyphenylacetone with NaBH3CN) To a well stirred solution of 31.0 g ethylamine hydrochloride in 110 mL MeOH there was added 6.6 g of 3,4-methylenedioxyphenylacetone (see under MDMA for its preparation) followed by 3.0 g sodium cyanoborohydride. Concentrated HCl in MeOH was added as required to maintain the pH at about 6 as determined with external, dampened universal pH paper. About 2 days were required for the reduction to be complete as determined by the final stabilization of the pH. The reaction mixture was added to 1 L H2O and made strongly acidic with an excess of HCl. After washing with 2x100 mL CH2Cl2 the aqueous phase was made basic with 25% NaOH, and extracted with 3x100 mL CH2Cl2. Removal of the solvent under vacuum yielded 8.3 g of a pale amber oil that was distilled at 85-100 °C at 0.2 mm/Hg. There was obtained 6.0 g of a water-white oil that was dissolved in 65 mL IPA and neutralized with 75 drops of concentrated HCl which produced crystals spontaneously. These were diluted with some 20 mL of anhydrous Et2O removed by filtration, washed first with IPA/Et2O (2:1), and then with Et2O. After air drying there was obtained 6.1 g of 3,4-methylenedioxy-N-ethylamphetamine hydrochloride (MDE) with a mp of 201-202 °C. Anal. (C12H18ClNO2) N.
DOSAGE: 100 - 200 mg.
DURATION: 3 - 5 h.
QUALITATIVE COMMENTS: (with 100 mg) There was a warm light all about me. And a gentle, almost alcohol-like, intoxication. The drug seems to change my state of awareness, but it does nothing else. The world is as intense or as dull as I choose to make it. At the 1.5 hour point I was clearly dropping, and an hour later yet, completely without residue.
(with 160 mg) The first effects were felt in forty minutes and I seemed to be completely there by the end of that first hour. There was an initial slightly dizzy intoxication, and then I felt very nice. A good intoxication, with maybe a little motor incoordination. There was absolutely no appetite at all. The next morning there was still some feeling of elation but I was still very relaxed. High marks for the quality of the experience.
(with 160 mg) Overall this was a wonderful experience. I felt that the effect was stronger and smoother than MDMA, but perhaps the group enhancement may be partly responsible. I felt definitely fewer physiological side-effects than with MDMA, particularly the urinating problem; although there was dehydration, there was less burning annoyance.
(with 160 mg) I was hard hit, to the extent that there was difficulty in verbalizing and following other people's thoughts. I entered the experience with some cold symptoms, and my sore throat disappeared. I felt quite intoxicated and tranquilized.
(with 200 mg) Very stoned. There was some nausea in the beginning of the experience. As it developed I found it very difficult to concentrate on what I was thinking or saying simply due to the extraordinary nature of coming on to this material. There is noticeable jaw-clenching and rice crispies in the ears. This is a meditative material not unlike MDMA except there are more difficulties in forming words. And there is a problem in focusing the eyes, what I want to call `eye-romp.' My anorexia was extremely long-lived-- perhaps a total of 72 hours. This may have been too high a dosage.
EXTENSIONS AND COMMENTARY: This immediate homologue of MDMA has a very similar chronology but requires a slightly larger dose. Another similarity is the occasional report of teeth clenching, especially following the use of supplemental dosages intended to extend the effects of the drug. These supplements have been explored in the 50 to 75 milligram range, usually at the two hour point. In one unpublished clinical experiment with MDMA, an extension was attempted at the 1 hour 45 minute point with MDE rather than with MDMA, to see if there was any change in the qualitative character of the experience. The effective time of intoxication was extended, but the group fell surprisingly quiet, with a drop in the usual urge to converse and interact.
The effects of MDE are similar in many ways to those of MDMA, but there are believable differences. The particular magic, and affective transference, does not appear to be there. There is a stoning intoxication, as there is with MDA, and there is a seemingly unrewarding aspect to the upping of the dosages, again similar to MDA, and the properties of unusually easy communication and positive self-viewing of MDMA seem to be absent. Maybe the "S" isomer would have these properties, and they are lost in the racemate due to something coming from a more potent "intoxicating" "R" isomer. The optical isomers have never been evaluated separately in man.
There are only two ways in which two drugs can interact to produce a result that is not obvious from the summing of their individual actions. One is the process of synergism, where two active materials are allowed to interact within a single individual and at one time, and the consequence of this interaction is different than that which would have been expected. The other is the process of potentiation, where only one drug is active, but the presence of the second (and inactive) drug enhances the observed action of the first. MDE seems to fall in the first category.
The "piggy-back" or "window exploitation" studes were first discovered and explored with MDE, and have subsequently been extended most successfully with MDMA. The earliest procedure used was to assay modest quantities of active materials at the drop-off period of MDE, to exploit the open and benign state that was present. Usually, only a fraction of the standard dosage of the following drug was necessary to evoke a full experience. In psychotherapy applications, this sequence has been frequently used with MDMA followed by a second material that has been chosen to modify and expand the opening that the MDMA produced.
With the placement of MDMA under legal control in 1985, MDE occasionally appeared in the illicit street trade. It had been called EVE, which carries some perverse logic in light of the nickname used occasionally for MDMA, which was ADAM. The term INTELLECT has been used for it as well, but there has been no apparent reason advanced for this. And a final note on nomenclature. An old literature use of the code MDE was for the compound 3,4-methylenedioxyethanol-amine. See the discussion on this under the recipe for DME.
I have been told of an analogue of MDE that has been synthesized, and explored by the researcher who synthesized it. It contains the N-trifluoroethyl group common to several pharmaceuticals such as Quazepam. The analogue is 3,4-methylenedioxy-N-(2,2,2-trifluoroethyl)amphetamine hydrochloride (mp 207-209 °C) which was made from 2,2,2-trifluoroethylamine and 3,4-methylenedioxyphenylacetone and sodium cyanoborohydride in methanol. The best final line for this compound is that it is "possibly active." The most heroic dosage schedule mentioned was a total of 500 milligrams, taken in three approximately equal portions over the course of five or six hours, with only a very mild intoxication and little or no sympathomimetic effects. And what little there might have been was quickly gone. A collection of totally unexplored N-substituted homologues and analogues of MDE is gathered at the end of the recipe for MDBZ.
Another direction that has been used to homologate the MDMA and MDE structure is with the length of the aliphatic chain that carries the phenyl ring and the amine function. RHS shows the two-carbon chain, "I" shows the amphetamine chain length, and MDE can be called ETHYL-I. The four-carbon chain is the RJS group, and this entire Muni-Metro concept is explained under METHYL-J.
| [ |
[Main Index] | [Forward |
| HTML and Design by Lamont Granquist & Erowid | Used by Erowid with permission of author |
|
[Plants & Drugs]
[Mind & Spirit]
[Freedom & Law]
[Arts & Sciences]
[Library]
[Search]
[About] (html and design © 1995-2009 Erowid.org. Please ask permission before publicly reproducing.) (Contents © respective copyright holders.) |
|---|

