
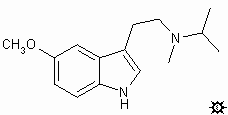
#40. 5-MEO-MIPT
TRYPTAMINE, N-ISOPROPYL-5-METHOXY-N-METHYL; INDOLE, 3-[2-(ISOPROPYLMETHYLAMINO)ETHYL]-5-METHOXY; N-ISOPROPYL-5-METHOXY-N-METHYLTRYPTAMINE; 3-[2-(ISOPROPYLMETHYLAMINO)ETHYL]-5-METHOXYINDOLE
|
| [3D .mol structure] |
DOSAGE : 4 - 6 mg, orally; 12 - 20 mg, smoked
DURATION : 4 - 6 hrs
QUALITATIVE COMMENTS : (with 1.5 mg, orally) "In 15 minutes I was already off of baseline, and by an hour it is unquestionably real. There is no visualization to music, but a general turned-on randiness. Dropping at an hour and a half, and out by three hours. Modest but real."
(with 4 mg, orally) "Up very fast, to a +2 in an hour. Absolutely no visuals, but over the next two hours an ease of interpretive fantasy, almost dream-like, and easy eroticism. Food tasted marvelous, but there was no appetite. Easy, normal sleep and good spirits in the AM."
(with 5 mg, orally) "Extremely bitter taste. Some stimulation and tingling at 10 minutes, and I am apprehensive at this rapid onset. There are no visual symptoms at all, but the stimulation of conceptual thought is intense. Depth perception is slightly altered and a very minor wave pattern can be noticed in the peripheral vision, but no major object or color distortion. Minor enhancement of auditory acuity. Philosophical concepts about this and other substances seem important. I wonder what the value of writing about, or attempting to describe, their effects really is. At the second hour, the effects are subsiding, and for another three hours there are tailings and insomnia, but I am able to eat normally. There is a certain amount of 'negativity' about this compound. The dose is satisfyingly small, but I wonder if it is worth it. Somewhat of a disappointment. Have no desire to experiment further."
(with 6 mg, orally) "Rapid development to 45 minutes, some shakes, uneven handwriting, and hints of time slowing. Extremely erotic. Full plateau at a +3 at the 2 1/2 hr. point, then a graceful and rather rapid drop. Easy, restful sleep. Absolutely no visuals or related sensory effects -- what does one call this stoned state? Very pleasant, music extremely acceptable, tactile extraordinary. I feel that higher dosages would not contribute anything more."
(with 12 mg, smoked) "I was able to take 4 or 5 puffs, and to hold off the onset until then, then I couldn't anymore. Powerful, tremendous rush, but all along maintaining body-ego awareness, unlike 5-MeO-DMT where the world appears to utterly dissolve. I was aware of doing a lot of groaning, writhing, shaking around; headphones and eyeshades kept it completely internal. Not too much visual, but lots of disorientation. Early on there was a lot of emotional lability, laughing, crying, 'Oh God' kinds of outbursts.
"At an hour and a half, I was down enough that I tried smoking the remainder of the bowl without much additional effect. A typical psychedelic afterglow and in the morning ate a big breakfast and felt essentially normal. In summary, I took too little, I was in a bad mood, and felt myself to be in a rushed environment. The experience seemed to me like a hybrid between CZ-74 and 5-MeO-DMT; the trippiness of the former and the rush (although not as intense) of the latter."
(with 20 mg, smoked) "Most all of it was smoked in about three or four inhalations before I felt it coming on so strongly that I lay down. Within less than a minute after I lay down, with my eyes closed, my visual field was filled with brilliant geometric patterned lines of different colors that were slowly moving. There were several sets of parallel and curved lines superimposed upon in each other. Soon after that, probably within a minute or two, I became extremely disoriented from my normal sense of being a person in a body; I was lost in an undifferentiated mass of feeling and non-specific sensation. It was similar to the overwhelming feeling of 5-MeO-DMT in quality and, as with that material, the peak phase lasted less than 30 minutes.
"Then I began to think more coherently, but intense waves would return every 5 to 15 minutes. In between my perception and thinking would be fairly normal, but with the waves I would be swept up in imagery or memories heavily laden with emotional content. After two hours I was joined by my wife. We spent some very intimate time together, and I remember asking her who she was and she replied, 'Your wife.' This was very powerful to me since I did not really know what it meant, except that it seemed to be the best combination of mother, lover and friend, and that it was an entirely new kind of relationship that we would be creating for the rest of our lives together.
"After three or four hours, the waves had virtually stopped, and I remained oriented to the present and my immediate surroundings. I stayed under a mild influence until I ate supper, around 7 hours. I felt tired, had trouble falling asleep, but awoke refreshed."
EXTENSIONS AND COMMENTARY : In my lecturing at the University, every couple of years or so some student uses the term "more unique than" or "relatively unique." This immediately triggers a reflex response from me, to emphasize the simple definition that something that is unique is something that is one of a kind, and that all one-of-a-kind things are different from all other one-of-a-kind things. All drugs are unique. Every drug is different from all other drugs. 5-MeO-MIPT is unique.
The last two entries in the "Qualitative Comments" section are longer than usual, but even at that they have been trimmed down from reports sent to me that were each of over three pages in length. A thread common to each of them, is the comparison of the effects of smoking 5-MeO-MIPT to those from smoking 5-MeO-DMT. The speed of onset, the intense depersonalization and loss of immediate contact with one's surroundings, the impressive recall of early memories and the significance of these memories, make the drugs appear similar to one another. And the fact that they are of similar potency when smoked (5-MeO-DMT is perhaps a tad more potent) makes the relationship more comfortable. And then, with the eye of a chemist making further comparisons, the whole structure-activity relationship falls into place. The formulae are identical, except that one of the N-methyl groups of 5-MeO-DMT is extended by a couple of carbon atoms, to an isopropyl group in 5-MeO-DIPT. They are almost the same. They have almost the same action when smoked. They are "unique and similar" and together they appear to be quite different from the rest of the pack. Nope, that is just not so! They are totally different from one another.
All you need to do, to see that clearly, is to look at that one additional observation involving oral activity. This drug, 5-MeO-MIPT, is several times more potent when taken orally than it is when smoked. 5-MeO-DMT is much less active orally than when it is smoked. As a matter of fact, it is not active at all when taken orally. No active oral level has ever been found. What a rich area for speculation. Preferential metabolism? First pass goings-on? Chemical change from pyrolysis in the pipe? Different receptors? Lipophilicity? I am reminded of the quote from Mark Twain; "I like science because it gives one such a wholesome return of conjecture from such a trifling investment of fact."
Might the observations of the remaining oxygen-substituted MIPT's provide additional clues? There are four possible mono-methoxylated MIPT's; all have been synthesized and all have been explored in man. The 4-methoxy-isomer was of modest activity and deserves, and has received, a recipe of its own. The 5-methoxy- isomer is the one described here, and is extremely potent (orally, but less so parenterally). But as one goes to the 6- and 7-positional isomers, the two remaining positions, the psychopharmacological activity seems to be lost. This is a humorous reminder of the British idiom, to be at 6-'s and 7-'s about something.
6-MeO-MIPT was made from the corresponding indole, by reaction with 2-nitroethyl acetate, the resulting 3-nitroethylindole catalytically hydrogenated to 6-MeO-T which was converted to the N-benzyloxycarbonyl derivative. This was reduced to 6-MeO-NMT which was in turn reductively coupled with acetone to provide 6-MeO-MIPT with a melting point of 89-91 °C and in an overall yield of 9%. MS (in m/z): C5H12N+ 86(100%); indolemethylene+ 160 (7%); parent ion 246 (4%). In human trials there was one report of some kind of neurological twinge at a 16 milligram level, but nothing else at trials of up to 50 milligrams and it has been shelved as being inactive.
The isomeric 7-MeO-MIPT was synthesized by the exact same five-step reaction sequence starting with 7-methoxyindole, in an overall yield of 24%. The actual reaction conditions for this conversion are detailed in the recipe for 4-MeO-MIPT. The mp of 7-MeO-MIPT was 72-73 °C and its MS (in m/z): C5H12N+ 86 (100%); indolemethylene+ 160 (5%); parent ion 246 (4%). Gas chromatographic analysis indicated that the product was only 80% pure, and three of the impurities were identified. One was 7-MeO-NIPT with MS (in m/z): C4H10N+ 72 (100%); indolemethylene+ 161/160 (13, 8%); parent ion 232 (4%). Another was 7-MeO-DMT with MS (in m/z): C3H8N+ 58 (100%); indolemethylene+ 160 (6%); parent ion 218 (9%). The third was 7-MeO-NMT with MS (in m/z): C2H6N+ 44 (100%); indolemethylene+ 161/160 (82, 39 %); parent ion 204 (5%). These three impurities represented approximately 5%, 3%, and 4%, resp., of the isolated product's final weight. It showed something going on at 20 mg orally with perhaps a little distortion in the visual field. And, separately, at 70 milligrams orally there might have been a light-headedness after a few minutes. Nothing more. It, too, has been given the kiss of death by being declared inactive at the 50 milligram level.
The last of the Mohicans, the tribe of compounds with the remarkably potent, orally active, N-methyl-N-isopropyl system on the tryptamine nitrogen atom, was the dimethoxy analogue with both the 5- and the 6-positions occupied with methoxy groups. This specific compound has its own recipe as it raises specific questions that deserve direct attention. The very close relative with the methylenedioxy group at this 5,6-location also has a separate recipe.
Two final laments. Remember that all these beautiful compounds are unique. Why do they behave the way they behave? I have no idea and there is never enough data to explain everything. I hate the fact that the word data is plural. But singular or plural keep collecting it (them), and keep trying to make sense of everything. And, a small point from my infancy. The Last of the Mohicans was one of the very first books I read, and I had very innocently accepted the footware of these Indians as being the metaphor for the people themselves. I had seen that title as having been, "The Last of the Moccasins." This is a pair of words that I still interchange without any defense, along with shoulder and soldier, avatar and atavar, and especially annoying when lecturing, irrelevant and irreverent.
| [ |
[Main Index] | [Forward |

