
#154 4-TIM
4-THIOISOMESCALINE; 2,3-DIMETHOXY-4-METHYLTHIOPHENETHYLAMINE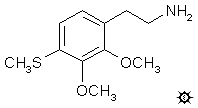
|
| [3D .mol structure] |
A solution of 1.2 g LAH in 20 mL anhydrous THF was cooled to 0 °C under He and stirred. There was added, dropwise, 0.8 mL of 100% H2SO4, followed by 0.9 g of 2,3-dimethoxy-4-methylthio-beta-nitrostyrene dissolved in 20 mL THF. Stirring was continued for a few min as the reaction returned to room temperature, and then it was heated to a reflux for 5 min on the steam bath. The reaction was cooled again, EtOAc was added to destroy the excess hydride, followed by 25% NaOH added dropwise until a white granular precipitate was obtained. This was removed by filtration, and the filter cake was washed with 2x35 mL Et2O. The filtrate was extracted into 50 mL dilute H2SO4 which was washed with Et2O and, in turn, made basic again and extracted with 2x50 mL CH2Cl2. The extracts were pooled, and the solvent removed under vacuum to give a residue of crude product. This distilled cleanly from 100-115 °C at 0.3 mm/Hg yielding 0.45 g of a clear white oil. This was dissolved in 6 mL IPA, neutralized with 5 drops of concentrated HCl, and diluted with 25 mL anhydrous Et2O. There was a deposition of white solids which were removed by filtration, washed with Et2O, and air dried. The 2,3-dimethoxy-4-methylthiophenethylamine hydrochloride so obtained (4-TIM) weighed 0.3 g and contained a molecule of H2O of crystallization. The mp was 212-213 °C. Anal. (C11H18ClNO2SaH2O) C,H,N.
DOSAGE: greater than 160 mg.
DURATION: unknown.
QUALITATIVE COMMENTS: (with 160 mg) Everything seemed normal. Pulse was under 80, there was nothing with eyes-closed, my appetite was normal. The compound was completely inactive.
EXTENSIONS AND COMMENTARY: There has been much noise made about the effectiveness of an unusual substitution group at the 4-position of the phenethylamine molecule. Here is a methylthio group at this position, and it is an inactive compound. I was just a little bit surprised.
| [ |
[Main Index] | [Forward |

